The Histology facility underpins the research activities of a large number of both basic and translational research groups. It allows the adoption of tissue-based experimental approaches to all research programmes.
The unit’s remit is to offer a full range of both routine and advanced histological services for oncology research.
The facility routinely processes, embeds and cuts numerous different tissue types in addition to organotypic assays, spheroids, agar plugs and cell pellets. Vibratome sections from fresh tissue (50–250μm) are also routinely prepared.
Cryostat sections are also routinely cut.
Routine equipment within the facility includes a Leica ASP6025 Tissue Processor and embedding station, a Leica Autostainer XL and a Thermo Cryostar NX70 cryostat. The unit also houses the Leica BondMax and BondRX immunohistochemistry platforms together with two Ventana Discovery units, which allow for both exceptional standardisation/quality and ultimate flexibility allowing sophisticated multiplexed labelling techniques to be incorporated into research programs.
This ultimately allows more information about the relationships between cells from different lineages on a single tissue sample and the gene expression information in the context of tissue/cell morphology to be analysed. The unit also houses the KOS Microwave Multifunctional Tissue Processor. This is ideally suited as a complement to our existing capabilities, where the multitasking nature of the instrument can enhance and accelerate many of the typical applications encountered. It allows for rapid tissue processing, bone marrow processing and decalcification, special stains, antigen retrieval, fixation and gross hardening.
Featured Publications

Mutant p53 induces SH3BGRL expression to promote cell engulfment
1st July 2025
In this paper, researchers show how mutant p53 cells drive the process of cell engulfment.
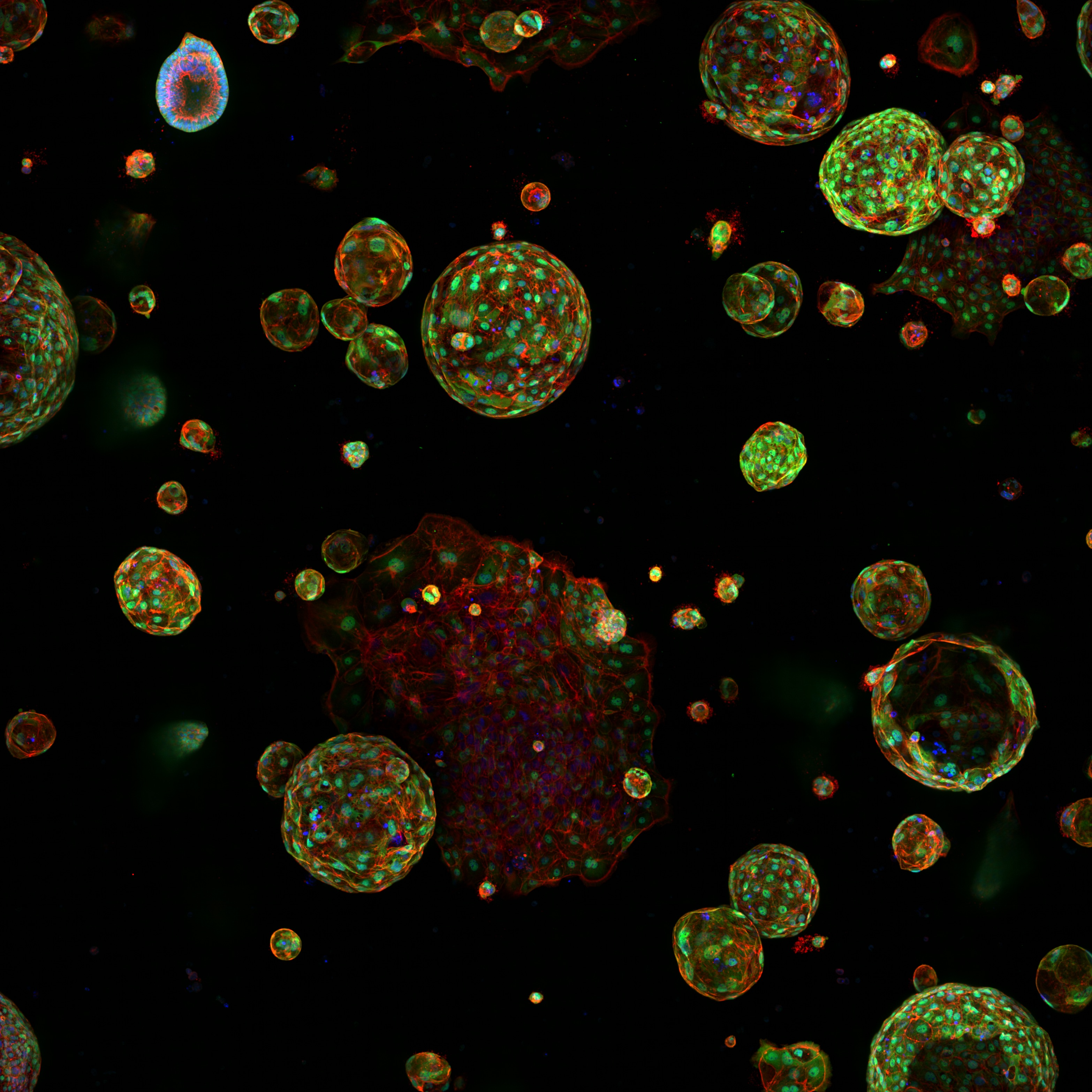
A microenvironment-inspired synthetic three-dimensional model for pancreatic ductal adenocarcinoma organoids
13th September 2021
Below et al. describe a fully synthetic hydrogel extracellular matrix designed to elicit key phenotypic traits of the pancreatic environment in culture.
Our Services
Today, with the continued development of the unit, the provision of histology services can be sub-divided into several areas. The breadth and depth of services offered, demonstrates how the unit underpins the research efforts of many groups.
Fixation, processing, microtomy & vibratome
No one fixative is ideal, and as result, the unit provides a selection of the most commonly used fixatives, together with advice for correct choice. Specimens are processed on the automated Shandon Excelsior or Leica ASP tissue processors using various schedules depending upon the size and nature of the tissue. Specialist tissue preparation, cut up and decalcification regimes can be employed if required.
Paraffin sections may be stained for morphological examination using the Leica autostainer XL. Unstained sections are used for downstream nucleic acid extraction, immunohistochemical analysis, in situ hybridisation, laser capture microdissection or spatial transcriptomics and proteomics.
The unit houses the Leica VT1200 vibratome. Without the freezing or processing of samples, the system allows for the sectioning of artifact free fresh tissue sections (up to 1000µm thick) to be used for culturing, immunohistochemistry or free-floating studies.

Cryotomy
Processing methods can have a deleterious effect on tissue constituents, and as a result, in some circumstances frozen sections may be the preferred method using the Thermo NX70 cryostat. Advice on all aspects of frozen tissue preparation and post fixation is available. The preparation of fixed cryoprotected samples is also routinely performed.

Special stains
Special stains allow for the demonstration of a variety of tissue constituents. Common requests include lipid, carbohydrate and cytoplasmic granule staining, together with various single and trichrome techniques for connective tissues and extracellular substances. Methods for specialised haematoxylin, nucleic acid and bone preparations are also available.

Brightfield and Fluorescence microscopy
The unit houses the Zeiss Axio Imager 2 upright microscope. With its various filter sets this allows for in-depth fluorescent and brightfield interrogation of tissue slides. With both a colour and black / white cameras the system can capture stunning quality images.

Nucleic acid extraction
The facility offers a range of extraction techniques, maximising both the quality and quantity of RNA / DNA captured. The resultant nucleic acids are quality checked as part of the service. Upstream, this service may incorporate either laser capture microdissection using the LMD600 or direct tissue macrodissection.

Spatial Technologies
The facility houses both the GeoMx DSP and Visium Cytassist platforms. The unit undertakes all the wet lab procedures up to and including hybridisation for these techniques. Optimisation of morphology markers for both the GeoMx DSP and phenocycler in the unit, is key to the success of these techniques.

Immunohistochemistry and In situ hybridisation
Immunohistochemistry is a technique employed by many groups within the Institute. The protocols can be automated or antibodies manually applied dependant on the downstream applications. Several epitope retrieval stations are also available.
Advice and support in all areas such as choice of fixation, epitope retrieval, method design / deployment and visualization is available.
Validation of novel antibodies, multiple chromogenic and fluorescent labelling panels, troubleshooting and interpretation of results are commonly undertaken.
Basescope and RNAscope are also offered as services. To interrogate the tissue fully these can be run as multiplex or single plex assays incorporating protein detection.

Get in touch
Our vision for world leading cancer research in the heart of Manchester
We are a leading cancer research institute within The University of Manchester, spanning the whole spectrum of cancer research – from investigating the molecular and cellular basis of cancer, to translational research and the development of therapeutics.
Our collaborations
Bringing together internationally renowned scientists and clinicians
Scientific Advisory Board
Supported by an international Scientific Advisory Board
Careers that have a lasting impact on cancer research and patient care
We are always on the lookout for talented and motivated people to join us. Whether your background is in biological or chemical sciences, mathematics or finance, computer science or logistics, use the links below to see roles across the Institute in our core facilities, operations teams, research groups, and studentships within our exceptional graduate programme.
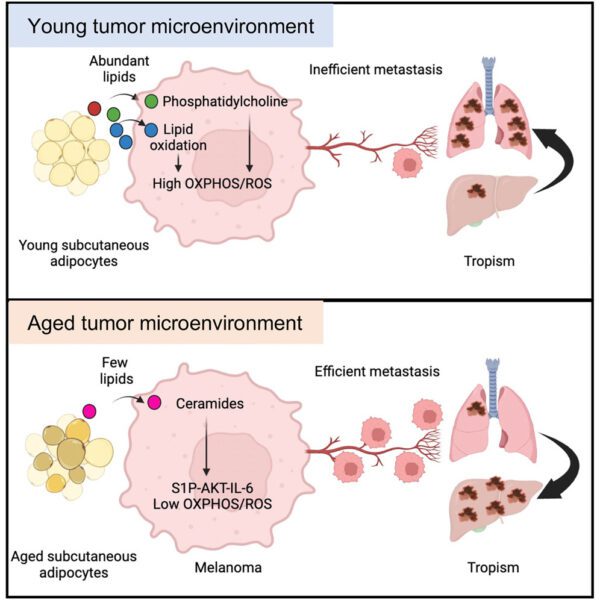
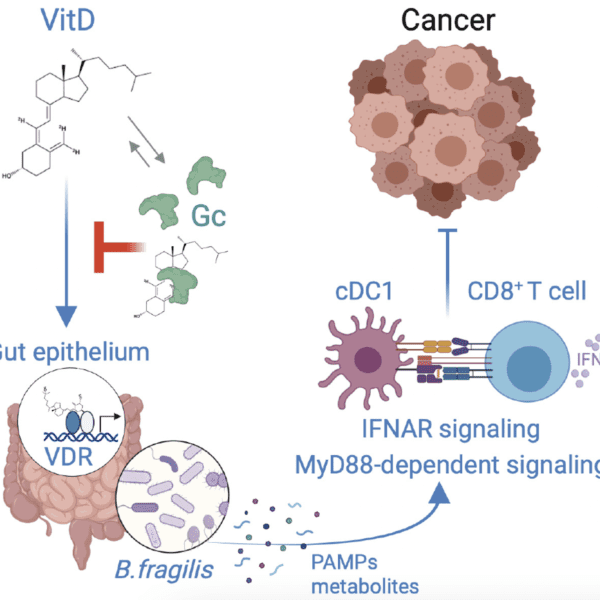


























A note from the Team Leader – Garry Ashton
The facility is housed together with other core facilities on the 4th floor of the new Paterson Building. The lab is subdivided into areas allowing the unit to offer a full range of both routine and advanced histological services for oncology research. The bespoke design of the area has allowed for range and complexity of the services offered to continue to grow and for a large number of both basic and translational research groups to adopt various tissue-based experimental approaches.