Editorial by Naomi van den Berg & Ben Evans
Paradoxes are fascinating contradictions of all our previous knowledge and experience – mysteries to solve.
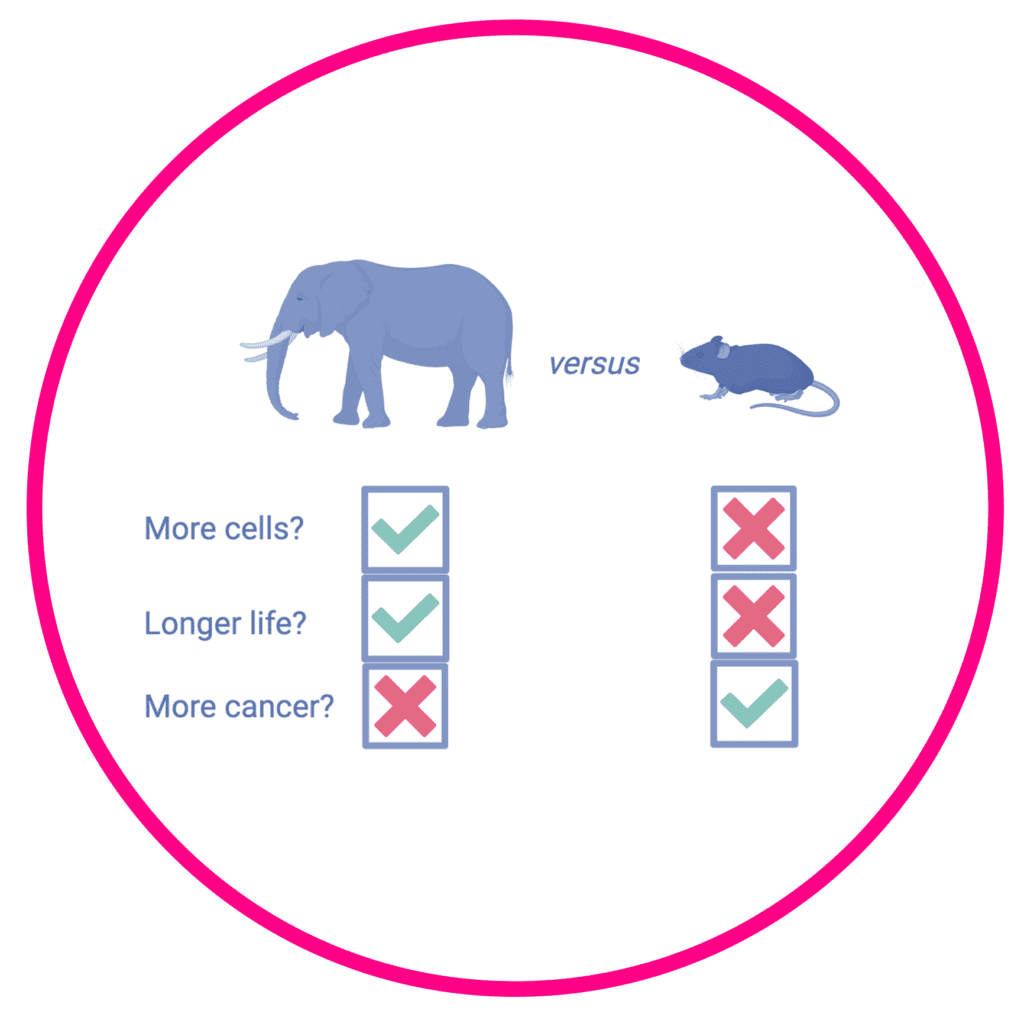
We see many such curious paradoxes in cancer. One is ‘Peto’s paradox’, the fact that large animals like elephants or whales get cancer less often than smaller animals (like we humans) do. This is despite having thousands of times more cells within their body than us, and therefore a theoretically higher chance of a cell becoming cancerous.
Another shows up in cancer rates between men and women. Even when we account for common lifestyle differences that might raise cancer risk, men still tend to develop more solid cancers in organs that both sexes share (so, not reproductive organs). You might expect those rates to be similar – but they aren’t.
If we can understand these paradoxes and their causes, we might be able to innovate new treatment options for cancer and improve the success rates of those that already exist. Our interest is in answering one particular cancer paradox, the ‘immune infiltration paradox’.
To do that, we need to dig down to the differences between cancer types.

The immune infiltration paradox we address in our latest research is a phenomenon linked to the prognosis of certain types of cancer.
Once a cell acquires changes that make it become cancerous, the cancer will grow within its local tissue and eventually form a tumour if not cleared up by the immune system.
How the tumour grows and spreads depends in part on how the immune system can detect and respond to it, a process that often involves immune cells entering the tumour, immune cell infiltration. There are many different types of immune cells that can enter the tumour, including T-cells: key cancer-killing immune cells.
In cancers such as melanoma or colorectal cancer, higher levels of T-cell infiltration are often associated with better patient outcomes. More infiltration usually means more effective control of the tumour.
But is this always the case? No.
There are a set of cancers that paradoxically have worse outcomes for the patient when T-cell infiltration into the tumour is high. These include cancers of the brain, eye and kidney. Why?
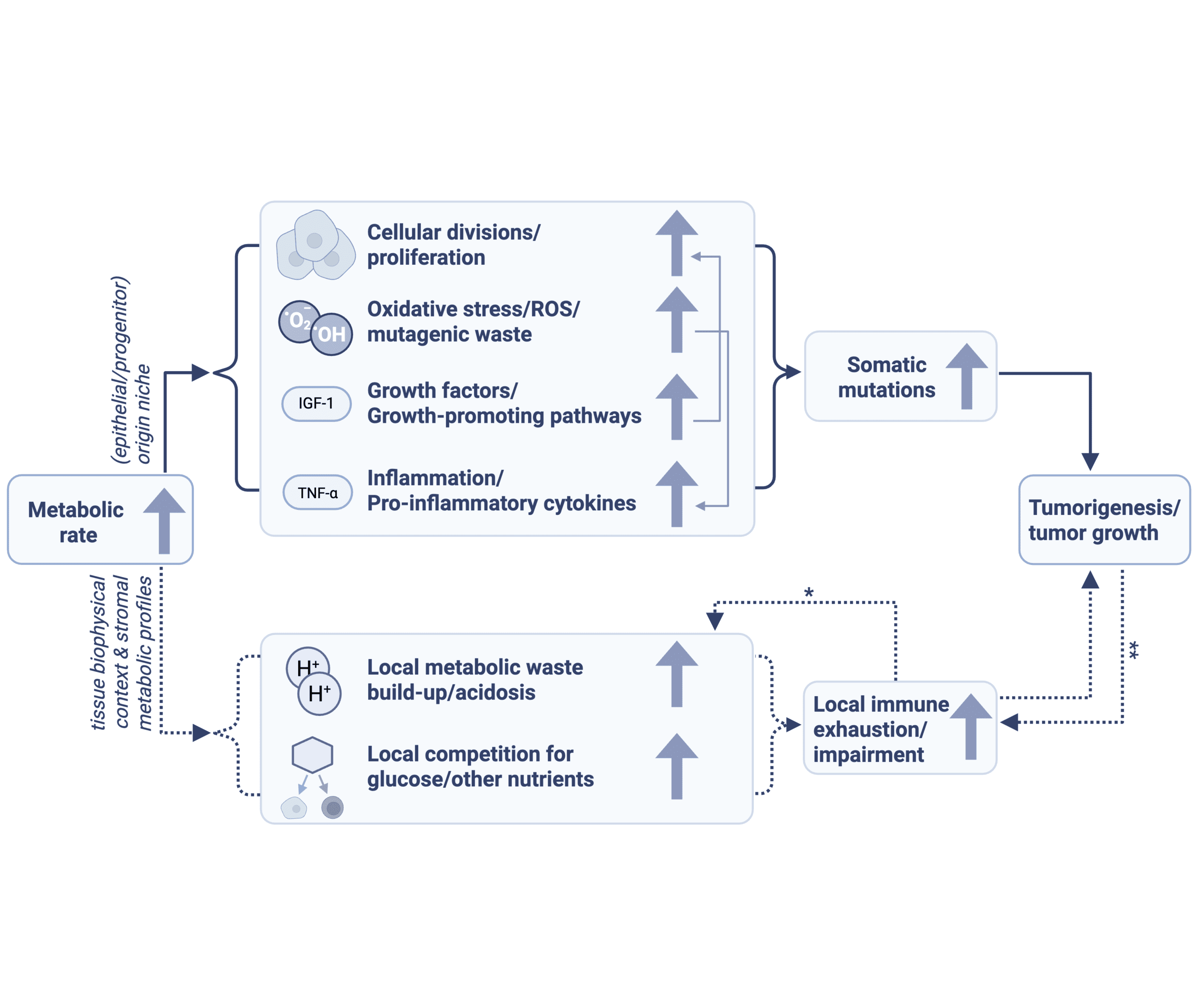
What does metabolism have to do with cancer?
Tumours within the body face very different environments depending on the tissue they’re in. What the kidney, brain and eye have in common is they’re all very metabolically intense tissues where huge amounts of energy are consumed.
Such energy consumption produces harmful waste in quantities that the body cannot entirely clear, making the local environment in that tissue relatively hostile compared to other parts of the body. For T immune cells entering the harsh tumour environment in these tissues, the challenging local conditions may prevent them operating as they should.
What’s important is that these conditions exist before the tumour even exists. It is the healthy tissue itself that is the metabolic constraint on the immune system.
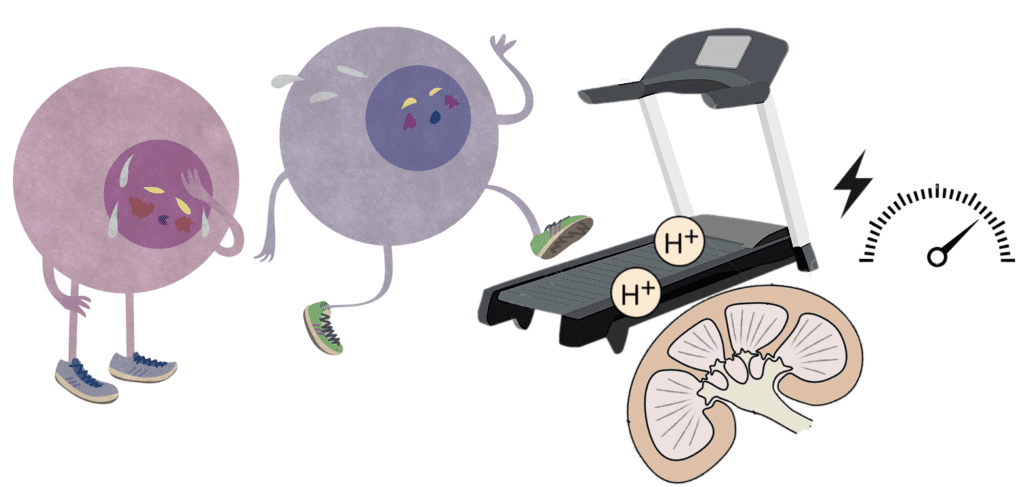
The treadmill model for immunometabolic gatekeeping
Let’s think about our local tissue as a treadmill, and our T-cells as enthusiastic runners trying to set a new personal best.
In a hostile tissue environment, the treadmill is covered in waste products that the T-cells must dodge. While normally a runner and a treadmill would be consuming two different types of fuel, in this case the T-cell and the tissue are fighting for the same limited resources. Imagine your treadmill was stealing your protein shake while you ran.
With no resource to fuel the runner and so many obstacles to dodge, they quickly get tired and secrete waste products of their own, which only clog up the treadmill with obstacles even further.
In some dense tissue structures the waste may be even harder to navigate, and the tumour could even harm the T-cells by slowing down their own ability to make energy. The runner has no chance of performing at their best, and in this analogy, the T-cells fail when operating at the edge of their limit.
We term this phenomenon ‘immunometabolic gatekeeping’, that tissue metabolism is one of the key features that conditions tumour immunity to the body immune system.
Improving cancer treatments and preventing cancer spread
Tackling immunometabolic gatekeeping is likely to be another tool in our arsenal to treat cancer. It can build on top of extensive research on tumour genetics and immune system checkpoints that have already led to improvements in survivability.
Immunotherapy treatments work by supporting the immune system to fight a cancer by increasing access for immune cells, turning on immune system triggers that have been disabled by a tumour, or by increasing the lethal potency of immune cells, but all of these methods might fail if the T-cells are constrained by the metabolic hostility of their local environment.
If we combine modern immunotherapy techniques with means of reducing the stress of high metabolic tissues on the immune system, we may be able to substantially increase treatment success rates and cancer survivability. The techniques for us to do this are numerous, from the usage of drugs to techniques such as moderating patient diet and exercise.
The next steps are to develop frameworks to test these ideas in an experimental and ultimately, a clinal setting. This includes exploring how interventions that target metabolism might improve the effects of immunotherapy in tumours that arose in high metabolism tissues.
Understanding and reducing the stress of high metabolism tissues on the immune system might help us improve cancer treatments and increase rates of survival.
Naomi van den Berg
Postdoctoral Fellow – Cancer Research UK Manchester Institute / Francis Crick Institute
Potential answers to cancer paradoxes
When we think back to other cancer paradoxes, immunometabolic gatekeeping may be a larger player than we realised. In Peto’s paradox, larger long-lived animals with lower metabolic rates may be able to deal with cancer more easily than us.
It may help to explain why men, with their higher average metabolism, have a higher risk of solid cancers than women, and explain other unanswered questions in cancer, such as childhood cancers or certain inherited cancers arising more commonly in tissues with higher metabolic rates.
Whether we’re thinking about improving immunotherapy or answering far broader cancer questions, considering metabolism as a major player in the role of immune function and cancer proliferation could be key to discoveries ahead.
Careers that have a lasting impact on cancer research and patient care
We are always on the lookout for talented and motivated people to join us. Whether your background is in biological or chemical sciences, mathematics or finance, computer science or logistics, use the links below to see roles across the Institute in our core facilities, operations teams, research groups, and studentships within our exceptional graduate programme.