Latest news & events from CRUK MI
All the latest news and updates from across the Institute
Latest news & events from CRUK MI
All the latest news and updates from across the Institute
Prizes, activities, events and more
See all the latest news from across our research teams, core facilities and staff. Use the search bar to select news items by category or date
News, Research Publication
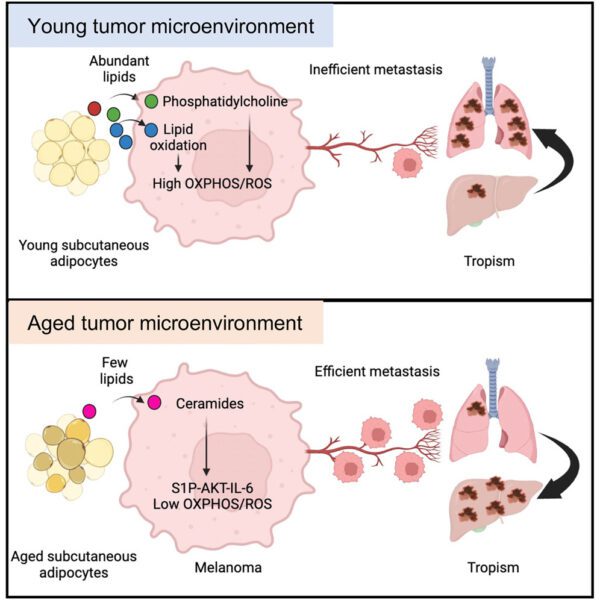
Can our metabolism explain cancer paradoxes?
16 April 2026
Topics & Tags
immunotherapy | kidney cancer | metabolism | publication | Cancer Immunology | Diet & Cancer | Immunotherapy
16 April 2026
News
New Research: tRNA modification triggers anti-tumour immunity
16 March 2026
New research has uncovered an unexpected way to stimulate the immune system against cancer: by subtly disrupting how tumour cells manufacture their proteins.
16 March 2026
Research Publication
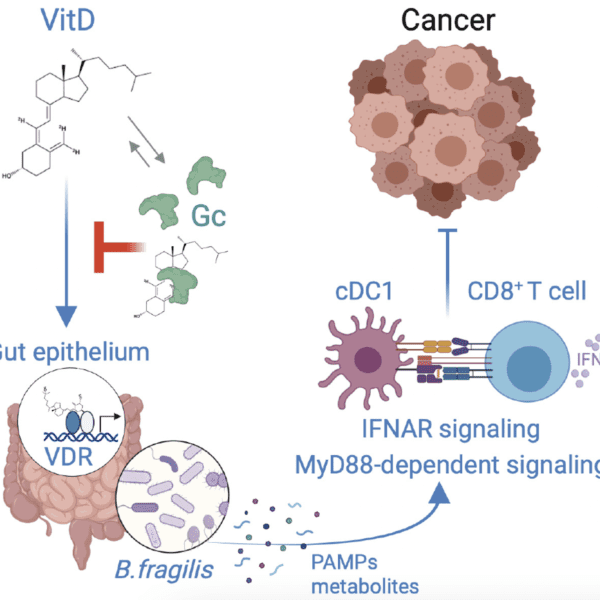
Cancer Inflammation and Immunity group publish exciting research in Cancer Discovery
20 October 2025
Topics & Tags
Immunotherapy | Skin Cancer
20 October 2025
News, Funding success, Staff News
Justin Loke appointed to Amit Patel Leukaemia Research Fellowship
28 August 2025
Topics & Tags
Amit Patel Leukaemia Research Fellowship | Blood Cancer | Leukaemia
28 August 2025
News, Research Engagement
Research Engagement Activities – How We Study Cancer
21 August 2025
We have developed research engagement activities which help explain how and why we use mice in some of our research, and new developments that can reduce or replacing mice in some experiments
Topics & Tags
AWERB | Culture of Care | Animal Models
21 August 2025
News, Funding success
Luciano Nicosia receives Blood Cancer UK Early Career Advancement Fellowship
6 August 2025
Dr Luciano Nicosia has been awarded a 3-year Early Career Advancement Fellowship with Blood Cancer UK.
Topics & Tags
Award | acute myeloid leukemia (AML) | Blood Cancer | Staff News
6 August 2025
Our Research
Our research spans the whole spectrum of cancer research from cell biology through to translational and clinical studies
Research Groups
Our research groups study many fundamental questions of cancer biology and treatment
Our Facilities
The Institute has outstanding core facilities that offer cutting edge instruments and tailored services from expert staff
Latest News & Updates
Find out all our latest news
Careers that have a lasting impact on cancer research and patient care
We are always on the lookout for talented and motivated people to join us. Whether your background is in biological or chemical sciences, mathematics or finance, computer science or logistics, use the links below to see roles across the Institute in our core facilities, operations teams, research groups, and studentships within our exceptional graduate programme.