Professor Caroline Dive
Director of the CRUK National Biomarker Centre | Small Cell Lung Cancer Biology Joint Group Leader
Caroline is Director of the aligned CRUK National Biomarker Centre, with research spanning tumour biology, preclinical pharmacology, biomarker discovery, biomarker assay validation and clinical qualification to regulatory standards, bioinformatics, biostatistics and most recently, digital clinical trials. Caroline is also a Senior Group Leader, jointly running the Small Cell Lung Cancer Biology group with Kathryn Simpson at the CRUK Manchester Institute. From 2021 to 2025, Caroline was also Interim Director of the Manchester Institute.
About Professor Caroline Dive
After completing her PhD studies in Cambridge, Professor Caroline Dive moved to Aston University’s School of Pharmaceutical Sciences in Birmingham where she established her own group studying mechanisms of drug induced tumour cell death, before moving to The University of Manchester to continue this research.
Caroline was awarded a Lister Institute of Preventative Medicine Research Fellowship before joining the CRUK Manchester Institute in 2003.
She is Director of the CRUK National Biomarker Centre, with research spanning tumour biology, preclinical pharmacology, biomarker discovery, biomarker assay validation and clinical qualification to regulatory standards, bioinformatics, biostatistics and most recently, digital clinical trials. From 2021 to 2025 she was Interim Director of the Manchester Institute and continues to co-lead the Small Cell Lung Cancer group within the Institute.
Caroline was awarded the Pasteur-Weizmann/Servier International Prize in 2012 for her Biomarker Research, the AstraZeneca Prize for Women in Pharmacology in 2016 and was presented with the 2019 Heine H. Hansen Lectureship Award by the International Association for the Study of Lung Cancer (IASLC). She is an elected Fellow of the Academy of Medical Sciences (2015), Fellow of the British Pharmacological Society (2012) and Fellow of the European Academy of Cancer Sciences (2011). In 2017, Caroline was awarded Commander of the Order of the British Empire (CBE) for her services to cancer research. Caroline became an elected member of EMBO (2020), received the first inaugural Johann Anton Merck Award in recognition for exceptional contributions to the field of preclinical oncology (2020), and was the recipient of the Mary J. Matthews Pathology/Translational Distinguished Service Award by IASLC (2021). Caroline was the President of the European Association for Cancer Research (2020 – 2022).
Qualifications
- PhD in Experimental Cancer Chemotherapy | 1988 | MRC Clinical Oncology, Cambridge
- Pharmacy Pre-Registration Training | 1985 | Eastbourne District General Hospital
- BPharm 1st Class Hons | 1984 | School of Pharmacy, University of London
Interests
- Small Cell Lung Cancer
- Cancer of Unknown Primary
- ctDNA liquid biopsy biomarkers
Research Projects
Publications
- MANIFEST: Multiomic platform for cancer immunotherapy
- Functional characterisation of the ATOH1 molecular subtype indicates a pro-metastatic role in small cell lung cancer
- A human model to deconvolve genotype-phenotype causations in lung squamous cell carcinoma
- A biobank of small cell lung cancer CDX models elucidates inter- and intratumoral phenotypic heterogeneity
- Lineage Plasticity in SCLC Generates Non-Neuroendocrine Cells Primed for Vasculogenic Mimicry
Why I work at CRUK MI
““I joined the Institute in 2004 to drive forward my basic discovery program of research on the regulation of drug-induced apoptosis in tumour cells. I quickly realised the enormous opportunity here adjacent to The Christie Hospital and, with encouragement from former Director Prof Nic Jones, I set about developing a cancer biomarker centre. I met thoracic oncologist Dr Fiona Blackhall during those early years, and she introduced me to the urgent clinical challenge of Small Cell Lung Cancer. We started our longstanding collaboration with the discovery of plentiful circulating tumour cells in the blood of patients with SCLC. Fast forward 20 years, I remain committed to research on mechanisms of chemoresistance and metastasis within Cancer Research UK Manchester Institute to improve outcomes in Small Cell Lung Cancer and now I direct the Institute-aligned Cancer Research UK National Biomarker Centre. The Institute is the perfect spot for translational research so I never left!””
Visit research group
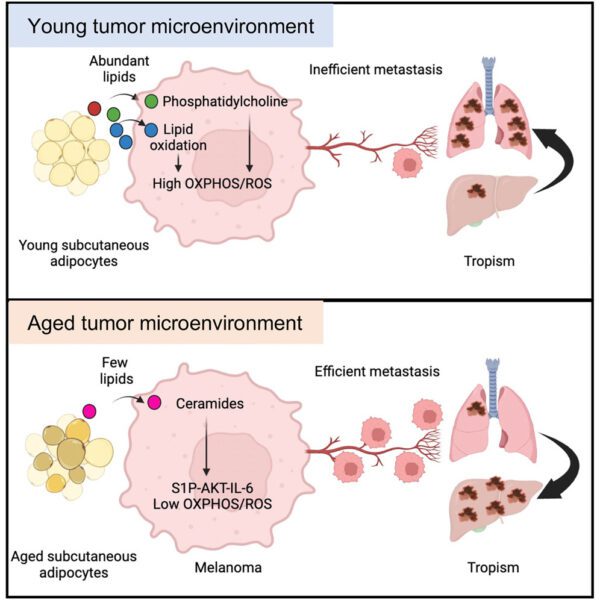
Small Cell lung Cancer (SCLC) is the most aggressive type of lung cancer and represents ~15% of all lung cancer cases with median survival of less than one year. SCLC is rapidly proliferating, highly metastatic and quickly develops chemotherapy resistance. Obtaining tumour biopsies is challenging and lack of molecular understanding of tumour heterogeneity historically prevented development of personalised therapy approaches. In 2014, our lab pioneered a preclinical model of SCLC known as a CDX (Circulating Tumour Cell (CTC) patient-derived explant) that capitalises on the high CTC burden of in patients. When isolated from a 7.5 ml blood draw, CTCs are engrafted into immune-compromised mice where resultant tumours reflect the molecular and phenotypic features of the donor patient’s tumour. After a decade of model development, we now have a biobank of >65 CDX models that recapitulate the extensive molecular heterogeneity of this disease, comprise the spectrum of therapy responses of donor patients and mimic metastatic traits. A wealth of multi-omic data exist for CDX models that combined with spatial information (multiplex imaging and transcriptomics) is being used to understand SCLC plasticity and evolution through treatment. Our focus is on gaining a better understanding of acquired chemotherapy resistance and the discovery of mechanisms of metastasis (especially to liver and to brain) to define personalised approaches to treatment. Our basic discovery program links to the portfolio of SCLC liquid biopsy biomarker development and validation efforts in the CRUK National Biomarker Centre.
Get in touch
Our vision for world leading cancer research in the heart of Manchester
We are a leading cancer research institute within The University of Manchester, spanning the whole spectrum of cancer research – from investigating the molecular and cellular basis of cancer, to translational research and the development of therapeutics.
Our collaborations
Bringing together internationally renowned scientists and clinicians
Scientific Advisory Board
Supported by an international Scientific Advisory Board
Careers that have a lasting impact on cancer research and patient care
We are always on the lookout for talented and motivated people to join us. Whether your background is in biological or chemical sciences, mathematics or finance, computer science or logistics, use the links below to see roles across the Institute in our core facilities, operations teams, research groups, and studentships within our exceptional graduate programme.