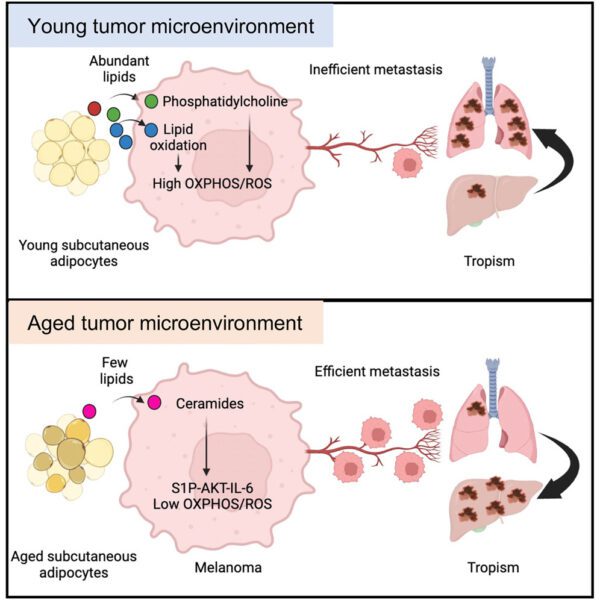
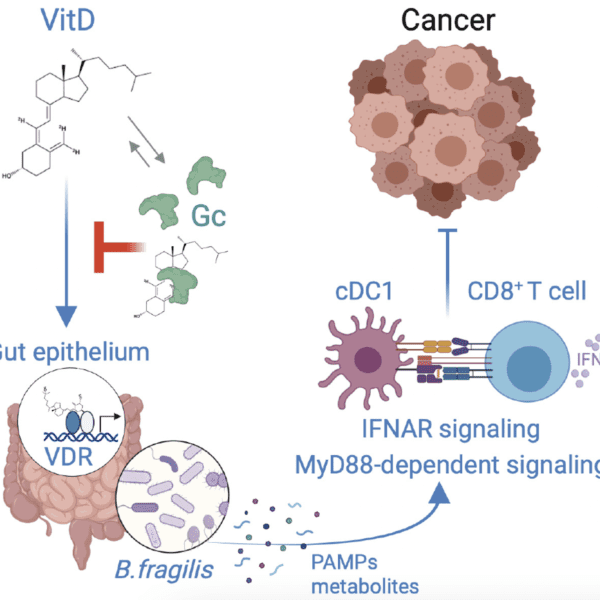
Our Research
Discover the cutting-edge research happening across our Institute
Our Research
Discover the cutting-edge research happening across our Institute
Research activities
Our research is driven by our innovative research groups who cover a wide range of topics and interests. Our PhD programme and ECR training support the development of researchers, while our Core Facilities provide infrastructure and expertise across a wide range of research areas.
Our Research Group Leaders

Samra Turajlić
Director & Senior Group Leader

Claus Jørgensen
Deputy Director & Senior Group Leader

Amaya Virós
Institute Fellow

Tim Somervaille
Senior Group Leader

Caroline Dive
Senior Group Leader

Santiago Zelenay
Senior Group Leader

Evangelos Giampazolias
Group Leader

Georges Lacaud
Senior Group Leader

Iain Hagan
Senior Group Leader

Mark Williams
Institute Fellow

Rob Bristow
Senior Group Leader

Kathryn Simpson
Team Lead

Sylvain Delaunay
Group Leader

William Hill
Institute Fellow

Justin Loke
Institute Fellow