Article highlights & insights
Background
The epigenetic factors KAT6A (MOZ/MYST3) and KMT2A (MLL/MLL1) interact in normal hematopoiesis to regulate progenitors’ self-renewal. Both proteins are recurrently translocated in AML, leading to impairment of critical differentiation pathways in these malignant cells. We evaluated the potential of different KAT6A therapeutic targeting strategies to alter the growth of KAT6A and KMT2A rearranged AMLs.
Methods
We investigated the action and potential mechanisms of the first-in-class KAT6A inhibitor, WM-1119 in KAT6A and KMT2A rearranged (KAT6Ar and KMT2Ar) AML using cellular (flow cytometry, colony assays, cell growth) and molecular (shRNA knock-down, CRISPR knock-out, bulk and single-cell RNA-seq, ChIP-seq) assays. We also used two novel genetic murine KAT6A models combined with the most common KMT2Ar AML, KMT2A::MLLT3 AML. In these murine models, the catalytic activity of KAT6A, or the whole protein, can be conditionally abrogated or deleted. These models allowed us to compare the effects of specific KAT6A KAT activity inhibition with the complete deletion of the whole protein. Finally, we also tested these therapeutic approaches on human AML cell lines and primary patient AMLs.
Results
We found that WM-1119 completely abrogated the proliferative and clonogenic potential of KAT6Ar cells in vitro. WM-1119 treatment was associated with a dramatic increase in myeloid differentiation program. The treatment also decreased stemness and leukemia pathways at the transcriptome level and led to loss of binding of the fusion protein at critical regulators of these pathways. In contrast, our pharmacologic and genetic results indicate that the catalytic activity of KAT6A plays a more limited role in KMT2Ar leukemogenicity, while targeting the whole KAT6A protein dramatically affects leukemic potential in murine KMT2A::MLLT3 AML.
Conclusion
Our study indicates that inhibiting KAT6A KAT activity holds compelling promise for KAT6Ar AML patients. In contrast, targeted degradation of KAT6A, and not just its catalytic activity, may represent a more appropriate therapeutic approach for KMT2Ar AMLs.
Background
The epigenetic factors KAT6A (MOZ/MYST3) and KMT2A (MLL/MLL1) interact in normal hematopoiesis to regulate progenitors’ self-renewal. Both proteins are recurrently translocated in AML, leading to impairment of critical differentiation pathways in these malignant cells. We evaluated the potential of different KAT6A therapeutic targeting strategies to alter the growth of KAT6A and KMT2A rearranged AMLs.
Methods
We investigated the action and potential mechanisms of the first-in-class KAT6A inhibitor, WM-1119 in KAT6A and KMT2A rearranged (KAT6Ar and KMT2Ar) AML using cellular (flow cytometry, colony assays, cell growth) and molecular (shRNA knock-down, CRISPR knock-out, bulk and single-cell RNA-seq, ChIP-seq) assays. We also used two novel genetic murine KAT6A models combined with the most common KMT2Ar AML, KMT2A::MLLT3 AML. In these murine models, the catalytic activity of KAT6A, or the whole protein, can be conditionally abrogated or deleted. These models allowed us to compare the effects of specific KAT6A KAT activity inhibition with the complete deletion of the whole protein. Finally, we also tested these therapeutic approaches on human AML cell lines and primary patient AMLs.
Results
We found that WM-1119 completely abrogated the proliferative and clonogenic potential of KAT6Ar cells in vitro. WM-1119 treatment was associated with a dramatic increase in myeloid differentiation program. The treatment also decreased stemness and leukemia pathways at the transcriptome level and led to loss of binding of the fusion protein at critical regulators of these pathways. In contrast, our pharmacologic and genetic results indicate that the catalytic activity of KAT6A plays a more limited role in KMT2Ar leukemogenicity, while targeting the whole KAT6A protein dramatically affects leukemic potential in murine KMT2A::MLLT3 AML.
Conclusion
Our study indicates that inhibiting KAT6A KAT activity holds compelling promise for KAT6Ar AML patients. In contrast, targeted degradation of KAT6A, and not just its catalytic activity, may represent a more appropriate therapeutic approach for KMT2Ar AMLs.
Institute Authors
Groups
Group leader
Research topics & keywords
Grants
The study was supported by Cancer Research UK (C5759/A20971 (G.L.), C5759/A27412 (G.L.), C19941/A31313 (M.S), Blood Cancer UK 19014 (G.L.), and an MSCA Postdoctoral Fellowship (658625, A.L.).
Meet the Research Team

Senior Group Leader
PhD Student
All publications
https://www.nature.com/articles/s42255-026-01514-y
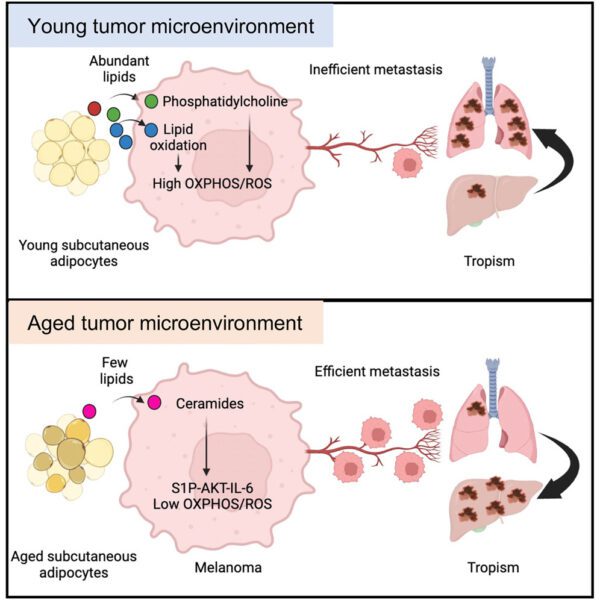
Tissue-specific fibroblast lipid cues impose the rate of epithelial cancer invasion
27 April 2026
Institute Authors (4)
Amaya Virós, Noah Palombo, Charlotte Russell, Claus Jørgensen
Research Group
Skin Cancer & Ageing
27 April 2026
https://www.cell.com/cancer-cell/fulltext/S1535-6108(26)00114-5
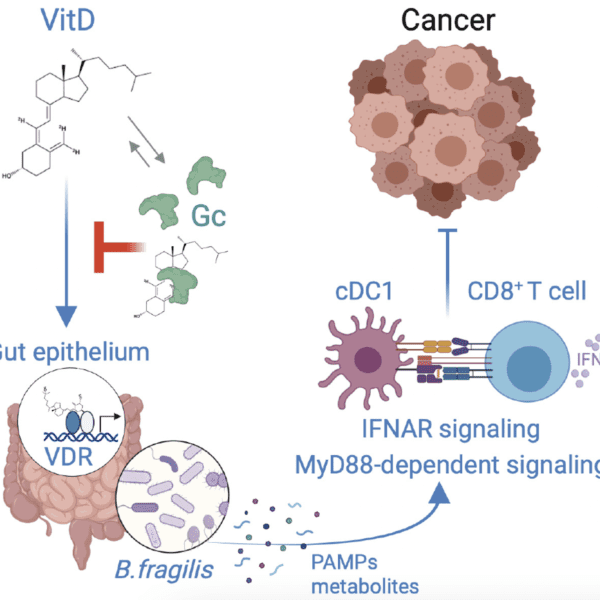
Immunometabolic gatekeeping: How tissue metabolism conditions tumor immunity
13 April 2026
Institute Authors (1)
Samra Turajlić
Research Group
Cancer Dynamics
13 April 2026
https://www.nature.com/articles/s41467-026-69964-2
Disruption of tRNA threonylation triggers RIG-I mediated anti-tumour immune response
25 February 2026
Institute Authors (1)
Sylvain Delaunay
Research Group
RNA Dynamics in Cancer
25 February 2026
https://doi.org/10.1038/s44161-025-00740-z
Single-cell profiling reveals three endothelial-to-hematopoietic transitions with divergent isoform expression landscapes
11 November 2025
Institute Authors (6)
Robert Sellers, John Weightman, Wolfgang Breitwieser, Natalia Moncaut, Michael Lie-a-ling, Georges Lacaud
Labs & Facilities
Computational Biology Support, Molecular Biology, Genome Editing and Mouse Models
Research Group
Stem Cell Biology
11 November 2025
https://doi.org/10.1136/jitc-2025-012527
Systemic immunosuppression from ultraviolet radiation exposure inhibits cancer immunotherapy
31 October 2025
Institute Authors (4)
Isabella Mataloni, Antonia Banyard, Garry Ashton, Amaya Virós
Labs & Facilities
Mass and Flow Cytometry, Histology
Research Group
Skin Cancer & Ageing
31 October 2025
https://aacrjournals.org/cancerdiscovery/article/doi/10.1158/2159-8290.CD-24-1224/766638/Glucocorticoids-Unleash-Immune-dependent-Melanoma
Glucocorticoids Unleash Immune-dependent Melanoma Control through Inhibition of the GARP/TGF β Axis
15 October 2025
Institute Authors (12)
Charles Earnshaw, Poppy Dunn, Shih-Chieh Chiang, Maria Koufaki, Massimo Russo, Kimberley Hockenhull, Erin Richardson, Anna Pidoux, Alex Baker, Richard Reeves, Robert Sellers, Sudhakar Sahoo
Labs & Facilities
Computational Biology Support, Visualisation, Irradiation and Analysis
Research Group
Cancer Inflammation and Immunity
15 October 2025
Our Research
Our research spans the whole spectrum of cancer research from cell biology through to translational and clinical studies
Research Groups
Our research groups study many fundamental questions of cancer biology and treatment
Our Facilities
The Institute has outstanding core facilities that offer cutting edge instruments and tailored services from expert staff
Latest News & Updates
Find out all our latest news
Careers that have a lasting impact on cancer research and patient care
We are always on the lookout for talented and motivated people to join us. Whether your background is in biological or chemical sciences, mathematics or finance, computer science or logistics, use the links below to see roles across the Institute in our core facilities, operations teams, research groups, and studentships within our exceptional graduate programme.